CHAPTER 6: Everyday Reactions
Science Understanding
- Describe chemical reactions using word equations identifying reactions and products
- Modeling chemical reactions in terms of rearrangement of atoms
- Recognize that the conservation of mass in a chemical reaction can be demonstrated by simple chemical equations
- Investigate reactions of acids with metals, bases and carbonates
- Investigating a range of different reactions to classify them as exothermic or endothermic
- Give example of how chemical reactions are important in both living and non living systems and involve energy transfer
- Compare respiration and photosynthesis and their role in biological processes
- Recognizing the role of oxygen in combustion reactions and comparing combustion with other oxidation reactions
- Describe how the products of combustion reaction affect the environment
Week 1
Week 2
Week 3
Week 4
Types of Chemical Reactions
Conservation of Mass
|
Balancing Chemical EquationsNow practice at home
|
PracticalsIn a chemical reaction the atoms are rearranged to form new substances.
The atoms in the reactants will be the same as the atoms in the products. The signs of a chemical reaction:
|
Week 5
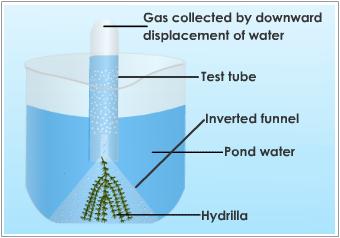
Reactions of LifeRespirationPhotosynthesis |
Endothermic/Exothermic
Endothermic = Energy moving in
Exothermic = Energy moving out
Is Photosynthesis exothermic or endothermic?
Is Respiration exothermic or endothermic? |
Practicals |
Week 6
RevisionHow to make revision notes:
1. Use key topic Headings
|
Chapter ReviewClick to get the Revision Scaffold. Save it and type in your summary information or use the topic headings to organise your own notes.
|
Test |